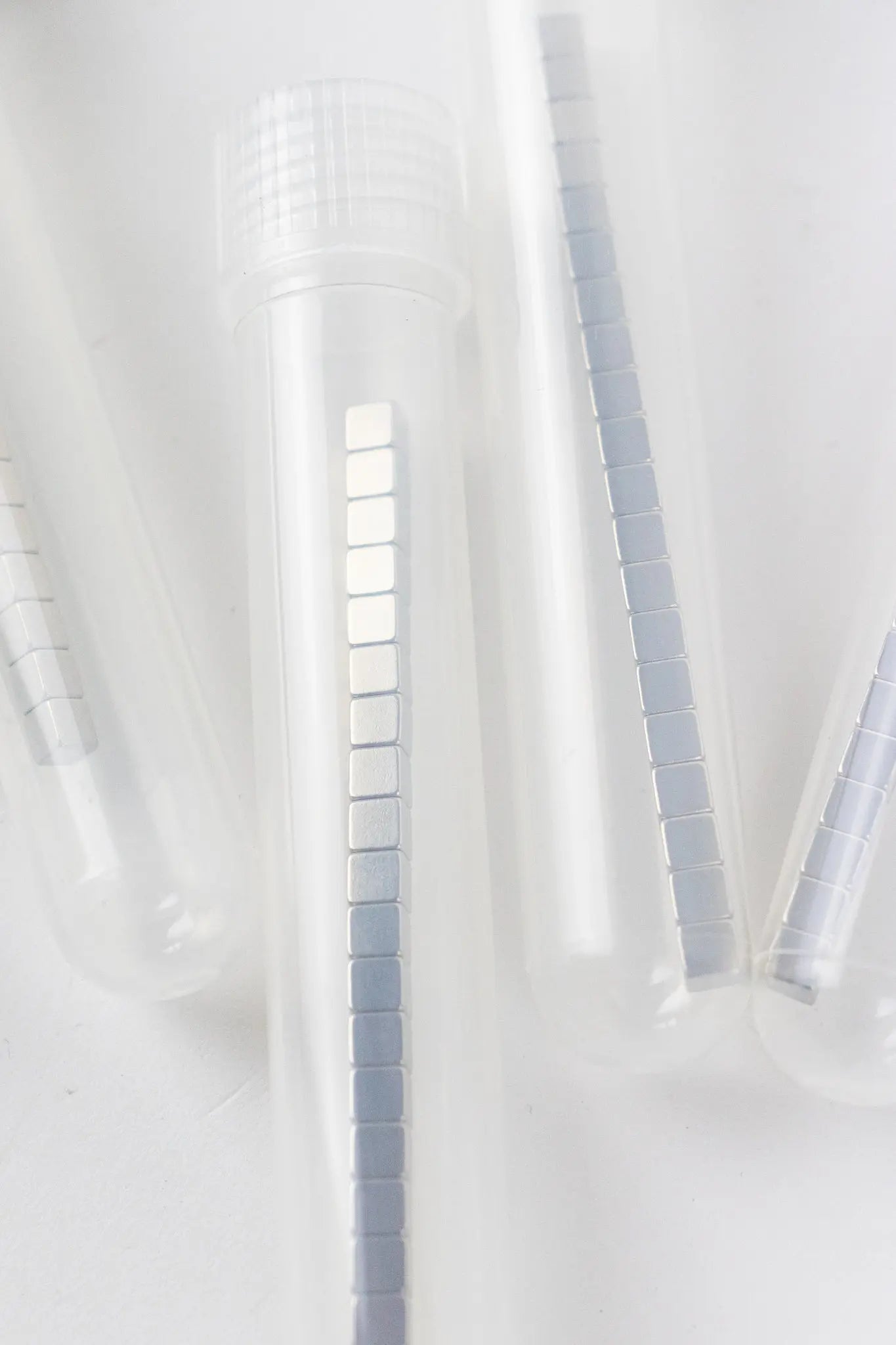
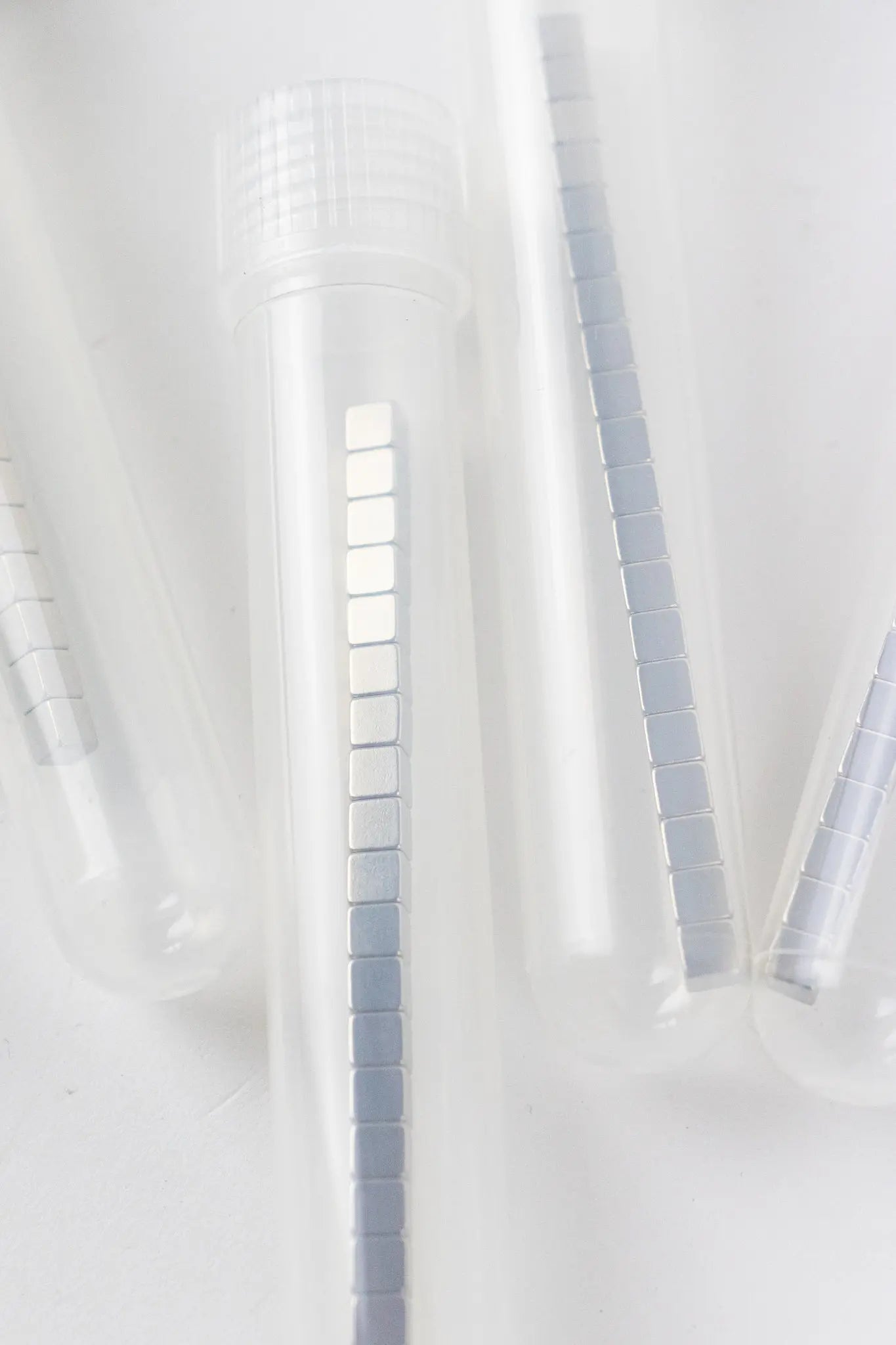
Tube of 20 Neodymium Magnets
Tube of 20 Neodymium Magnets
5.0 / 5.0
(5) 5 total reviews
 Item #020
Item #020
Our mighty 4mm cubic rare earth magnets are incredibly useful, mesmerizing to fiddle with, and a steal for only 5 bucks for a tube of 20.
We had these custom manufactured to the highest magnetization the factory could concentrate into the small space.
Made from a neodymium (Nd) alloy, these magnets each have about a 4lb pull (direct metal-to-metal contact). They are nickel coated since the magnetic material is a highly compressed powder.
*NOT A TOY. NOT FOR YOUNG CHILDREN. DO NOT SWALLOW*
More info:
Only a few elements are naturally magnetic at room temperature. Neodymium is not one of these elements; however, in the 1980s, General Motors found that an alloy of neodymium, iron, and boron (Nd2Fe14B) not only produces a magnet, but its crystal structure causes it to be an especially strong magnet.
Also called “rare-earth” magnets since neodymium is in the group of elements called rare-earths.
- Research Card
Matter Subscriber Price:
Couldn't load pickup availability




Collections containing this item:
Bestsellers
|
Items ≤ $20
|
Magnetics
|
Matter Items
|
Newest Listings
|
Physicsfun
|
Stemcell Catalog
|
Trinkets
|
AUTHENTICITY GUARANTEED
We only list 100% verified authentic items. We work with reputable collectors, and regularly consult with our network of scientists and experts.
-
Free Shipping
Orders ≥$50 qualify for free US shipping
Orders ≥$100 qualify for free global shipping -
Secure Payments
Pay how you like: credit card, PayPal, After Pay, Shop Pay, Venmo, Apple/Google/Meta Pay & crypto
-
Simple Returns
Change your mind? No problem. Enjoy easy returns within 30 days.
Amazing
i really like them there fun to play with.
I bought these to use with some magnetic sand, and they work wonderfully. I drop one down a thin test tube so I can wipe off the sand easily afterward, plus it shows well against the glass. Great value for 20 of them.
Just what I was looking for. These are nice strong little magnets. Came promptly. Love this shop!
This is a neat set of neodymium magnets that I have on my desk at work, they are a minor conversation starter but are also great for implanting inside 3D prints.
similar items_
WE TAKE SCIENCE SERIOUSLY
You deserve better than craft store science products. And we think learning is more impactful when you're holding a tangible piece of what you're learning about. That's why Stemcell exists.
We're dedicated to providing the best scientific products available—whether they're fragments of scientific importance, experimental activities, or just interesting things that scratch your curiosity itch.
With every new product launch, our list of new ideas gets longer rather than shorter. So check in often for our latest projects, and thanks for being a part of our endeavor to make the world a smarter place to live.
Believe in yourself; for everything else, there's science.
— TERRY MUDGE, FOUNDER & SCIENTIFIC DIRECTOR